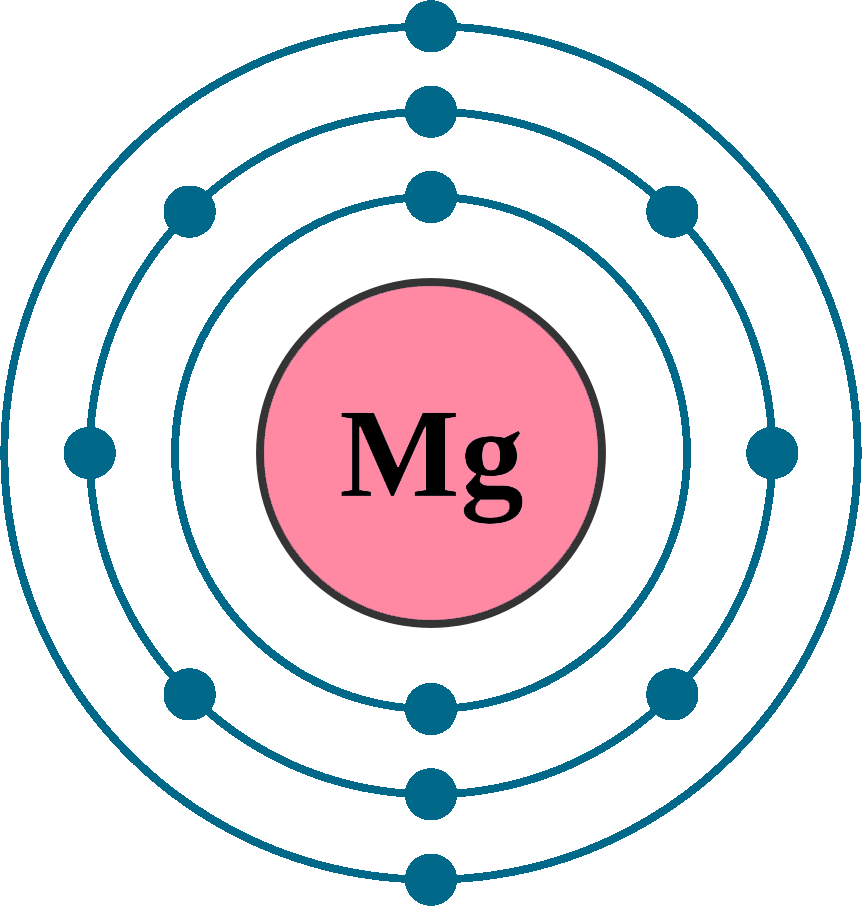
Magnesium Ion Have Electrons . The ground state electron configuration of magnesium is important in understanding. (a) since mg 2 + is a cation, its name is the name of the element it comes from plus the word ion. a magnesium atom has 2 electrons in its outer shell; electron configuration of magnesium is [ne] 3s2. Nitrogen’s position in the periodic table (group 15). in order to write the mg electron configuration we first need to know the number of electrons for the mg atom (there are 12 electrons). magnesium has two valence electrons in the 3s orbital. A magnesium atom can lose both outer electrons, so a magnesium ion has a charge. elements are organised into blocks by the orbital type in which the outer electrons are found. Possible oxidation states are +2. This would make it a. the symbol for the ion is mg 2 +, and it is called a magnesium ion. These blocks are named for the.
from www.newtondesk.com
Nitrogen’s position in the periodic table (group 15). elements are organised into blocks by the orbital type in which the outer electrons are found. the symbol for the ion is mg 2 +, and it is called a magnesium ion. magnesium has two valence electrons in the 3s orbital. These blocks are named for the. This would make it a. (a) since mg 2 + is a cation, its name is the name of the element it comes from plus the word ion. in order to write the mg electron configuration we first need to know the number of electrons for the mg atom (there are 12 electrons). The ground state electron configuration of magnesium is important in understanding. A magnesium atom can lose both outer electrons, so a magnesium ion has a charge.
magnesium electron configuration Newton Desk
Magnesium Ion Have Electrons These blocks are named for the. electron configuration of magnesium is [ne] 3s2. Possible oxidation states are +2. in order to write the mg electron configuration we first need to know the number of electrons for the mg atom (there are 12 electrons). a magnesium atom has 2 electrons in its outer shell; This would make it a. (a) since mg 2 + is a cation, its name is the name of the element it comes from plus the word ion. magnesium has two valence electrons in the 3s orbital. the symbol for the ion is mg 2 +, and it is called a magnesium ion. elements are organised into blocks by the orbital type in which the outer electrons are found. A magnesium atom can lose both outer electrons, so a magnesium ion has a charge. The ground state electron configuration of magnesium is important in understanding. Nitrogen’s position in the periodic table (group 15). These blocks are named for the.
From www.vectorstock.com
Symbol and electron diagram for magnesium Vector Image Magnesium Ion Have Electrons A magnesium atom can lose both outer electrons, so a magnesium ion has a charge. electron configuration of magnesium is [ne] 3s2. elements are organised into blocks by the orbital type in which the outer electrons are found. The ground state electron configuration of magnesium is important in understanding. (a) since mg 2 + is a cation,. Magnesium Ion Have Electrons.
From commons.wikimedia.org
FileElectron shell 012 Magnesium.svg Wikimedia Commons Magnesium Ion Have Electrons Nitrogen’s position in the periodic table (group 15). A magnesium atom can lose both outer electrons, so a magnesium ion has a charge. in order to write the mg electron configuration we first need to know the number of electrons for the mg atom (there are 12 electrons). magnesium has two valence electrons in the 3s orbital. . Magnesium Ion Have Electrons.
From guidemanualeruptivity.z14.web.core.windows.net
Magnesium Electron Configuration Diagram Magnesium Ion Have Electrons electron configuration of magnesium is [ne] 3s2. These blocks are named for the. Possible oxidation states are +2. elements are organised into blocks by the orbital type in which the outer electrons are found. The ground state electron configuration of magnesium is important in understanding. in order to write the mg electron configuration we first need to. Magnesium Ion Have Electrons.
From periodictable.me
Magnesium Electron Configuration (Mg) with Orbital Diagram Magnesium Ion Have Electrons Possible oxidation states are +2. the symbol for the ion is mg 2 +, and it is called a magnesium ion. Nitrogen’s position in the periodic table (group 15). A magnesium atom can lose both outer electrons, so a magnesium ion has a charge. magnesium has two valence electrons in the 3s orbital. elements are organised into. Magnesium Ion Have Electrons.
From mungfali.com
Magnesium Ion Electron Configuration Magnesium Ion Have Electrons magnesium has two valence electrons in the 3s orbital. in order to write the mg electron configuration we first need to know the number of electrons for the mg atom (there are 12 electrons). This would make it a. The ground state electron configuration of magnesium is important in understanding. A magnesium atom can lose both outer electrons,. Magnesium Ion Have Electrons.
From cezzhgjh.blob.core.windows.net
Magnesium Electrons at Marsha Dennis blog Magnesium Ion Have Electrons (a) since mg 2 + is a cation, its name is the name of the element it comes from plus the word ion. in order to write the mg electron configuration we first need to know the number of electrons for the mg atom (there are 12 electrons). Possible oxidation states are +2. These blocks are named for. Magnesium Ion Have Electrons.
From www.slideserve.com
PPT Atoms the building blocks PowerPoint Presentation, free Magnesium Ion Have Electrons magnesium has two valence electrons in the 3s orbital. Possible oxidation states are +2. (a) since mg 2 + is a cation, its name is the name of the element it comes from plus the word ion. in order to write the mg electron configuration we first need to know the number of electrons for the mg. Magnesium Ion Have Electrons.
From cecbfhcq.blob.core.windows.net
Atomic Number Of Magnesium Have at Wilson Turner blog Magnesium Ion Have Electrons magnesium has two valence electrons in the 3s orbital. a magnesium atom has 2 electrons in its outer shell; in order to write the mg electron configuration we first need to know the number of electrons for the mg atom (there are 12 electrons). (a) since mg 2 + is a cation, its name is the. Magnesium Ion Have Electrons.
From www.sciencephoto.com
Magnesium, atomic structure Stock Image C018/3693 Science Photo Library Magnesium Ion Have Electrons Nitrogen’s position in the periodic table (group 15). These blocks are named for the. This would make it a. in order to write the mg electron configuration we first need to know the number of electrons for the mg atom (there are 12 electrons). (a) since mg 2 + is a cation, its name is the name of. Magnesium Ion Have Electrons.
From www.animalia-life.club
Magnesium Electron Configuration Magnesium Ion Have Electrons a magnesium atom has 2 electrons in its outer shell; elements are organised into blocks by the orbital type in which the outer electrons are found. in order to write the mg electron configuration we first need to know the number of electrons for the mg atom (there are 12 electrons). This would make it a. A. Magnesium Ion Have Electrons.
From www.dreamstime.com
Electron of the Element Magnesium Stock Vector Illustration of Magnesium Ion Have Electrons This would make it a. A magnesium atom can lose both outer electrons, so a magnesium ion has a charge. magnesium has two valence electrons in the 3s orbital. These blocks are named for the. in order to write the mg electron configuration we first need to know the number of electrons for the mg atom (there are. Magnesium Ion Have Electrons.
From www.slideserve.com
PPT Electrons in atoms. PowerPoint Presentation, free download ID Magnesium Ion Have Electrons magnesium has two valence electrons in the 3s orbital. the symbol for the ion is mg 2 +, and it is called a magnesium ion. a magnesium atom has 2 electrons in its outer shell; The ground state electron configuration of magnesium is important in understanding. This would make it a. in order to write the. Magnesium Ion Have Electrons.
From slideplayer.com
SOL Review Game ppt download Magnesium Ion Have Electrons The ground state electron configuration of magnesium is important in understanding. Possible oxidation states are +2. electron configuration of magnesium is [ne] 3s2. These blocks are named for the. the symbol for the ion is mg 2 +, and it is called a magnesium ion. A magnesium atom can lose both outer electrons, so a magnesium ion has. Magnesium Ion Have Electrons.
From periodictable.me
Magnesium Electron Configuration (Mg) with Orbital Diagram Magnesium Ion Have Electrons elements are organised into blocks by the orbital type in which the outer electrons are found. These blocks are named for the. magnesium has two valence electrons in the 3s orbital. A magnesium atom can lose both outer electrons, so a magnesium ion has a charge. a magnesium atom has 2 electrons in its outer shell; Possible. Magnesium Ion Have Electrons.
From www.animalia-life.club
Magnesium Electron Configuration Magnesium Ion Have Electrons This would make it a. These blocks are named for the. (a) since mg 2 + is a cation, its name is the name of the element it comes from plus the word ion. magnesium has two valence electrons in the 3s orbital. in order to write the mg electron configuration we first need to know the. Magnesium Ion Have Electrons.
From valenceelectrons.com
How to Find the Valence Electrons for Magnesium (Mg)? Magnesium Ion Have Electrons Nitrogen’s position in the periodic table (group 15). elements are organised into blocks by the orbital type in which the outer electrons are found. magnesium has two valence electrons in the 3s orbital. electron configuration of magnesium is [ne] 3s2. in order to write the mg electron configuration we first need to know the number of. Magnesium Ion Have Electrons.
From circuitwiringhirsle77.z22.web.core.windows.net
Magnesium Dot Diagram Magnesium Ion Have Electrons These blocks are named for the. the symbol for the ion is mg 2 +, and it is called a magnesium ion. electron configuration of magnesium is [ne] 3s2. Possible oxidation states are +2. (a) since mg 2 + is a cation, its name is the name of the element it comes from plus the word ion.. Magnesium Ion Have Electrons.
From utedzz.blogspot.com
Periodic Table Magnesium Electron Configuration Periodic Table Timeline Magnesium Ion Have Electrons magnesium has two valence electrons in the 3s orbital. (a) since mg 2 + is a cation, its name is the name of the element it comes from plus the word ion. The ground state electron configuration of magnesium is important in understanding. Nitrogen’s position in the periodic table (group 15). These blocks are named for the. . Magnesium Ion Have Electrons.